Open Nav

Recently, Associate Researcher Qiu Pengfei, Researcher Shi Xun, and Researcher Chen Lidong from the Shanghai Institute of Ceramics, Chinese Academy of Sciences, in collaboration with Professor G. Jeffrey Snyder from Northwestern University in the U.S. and Professor Jürgen Janek from Justus Liebig University Giessen in Germany, conducted an in-depth analysis of the migration and precipitation mechanisms of mobile ions in quasi-liquid thermoelectric materials under external electric fields. Combining theoretical and experimental approaches, they proposed a thermodynamic stability criterion for determining whether "quasi-liquid" ions can precipitate out of the material, and also provided corresponding experimental characterization methods and techniques. On this basis, they suggested that introducing an interface featuring "ion blocking—electron conduction" can significantly enhance the operational stability of quasi-liquid thermoelectric materials under strong electric fields or large temperature differences. This research holds significant implications for the practical application of quasi-liquid thermoelectric materials. The relevant findings have been published in Nature Communications (DOI: 10.1038/s41467-018-05248-8). The equipment independently developed by the research team and some of the measurement results have been published in the Journal of Inorganic Materials (Vol. 32, 2017, 1337–1344), and a Chinese invention patent has been filed.
Thermoelectric energy conversion technology leverages the Seebeck and Peltier effects of semiconductor materials to directly convert thermal energy into electrical energy, holding significant and broad application prospects in areas such as power generation from industrial waste heat and automotive exhaust waste heat. However, constrained by the long-range order of their structures, conventional crystalline thermoelectric materials exhibit a minimum limit (the lowest lattice thermal conductivity) for lattice thermal conductivity, which restricts the room for continuous optimization of thermoelectric performance. To address this bottleneck, since 2012, the thermoelectric team led by Researcher Li-Dong Chen and Researcher Xun Shi at the Shanghai Institute of Ceramics has proposed introducing ions with "liquid-like" characteristics into solid-state materials to reduce thermal conductivity and optimize thermoelectric performance. They have successfully broken through the limitation of lattice thermal conductivity in solid-state glasses or crystalline materials, thereby discovering a new class of high-performance (ZT~2.0 at 1000 K) liquid-like thermoelectric material systems characterized by "phonon liquid–electron crystal" features (Nat. Mater. 2012, Adv. Mater. 2013&2014&2015&2017, Energ. Environ. Sci. 2014&2017, npj Asia Mater. 2015, etc.), making this an emerging hot topic in the field of thermoelectric materials in recent years. Nevertheless, the metallic cations with "liquid-like" characteristics in these liquid-like thermoelectric materials (such as Cu2-δSe, Ag9GaSe6, Zn4Sb3, etc.) tend to migrate over long distances under the influence of electric or temperature fields and subsequently precipitate out, leading to poor operational stability and limiting their practical applications. Therefore, understanding the migration processes and physical mechanisms of ions in liquid-like thermoelectric materials and thereby enhancing their operational stability is crucial for bringing new high-performance liquid-like thermoelectric materials toward practical applications.
The research team found that under the influence of an external field, metallic cations (such as Cu, Ag, and Zn) in liquid-like thermoelectric materials will undergo long-range directional migration from one end of the sample to the other, creating an ion concentration gradient. However, metallic cations will only precipitate out of the material and transform into metallic elements when their chemical potential at high concentrations is equal to or higher than the chemical potential of the corresponding metallic element itself, thereby triggering material decomposition. Consequently, each liquid-like thermoelectric material has a thermodynamic stability limit; ion precipitation and material decomposition will occur only when the external field is strong enough to push the material beyond this limit. Otherwise, the liquid-like thermoelectric material will behave similarly to conventional crystalline thermoelectric compounds, maintaining excellent stability and thermoelectric performance under the influence of an external field. Based on derivations using electrochemical formulas, the team discovered that the specific value of this thermodynamic limit can be determined by the maximum applied voltage that the material can withstand without undergoing decomposition—known as the critical voltage. The critical voltage is a characteristic parameter that is independent of the material's dimensions and depends solely on its intrinsic chemical composition and the ambient temperature.
To experimentally demonstrate the existence of thermodynamic stability limits in liquid-like thermoelectric materials, the team independently developed an instrument for quantitatively characterizing the operational stability of such materials. Under constant-temperature and fixed temperature-difference conditions, they successfully measured the critical voltages of a series of Cu2-δ(S,Se) liquid-like thermoelectric materials by using changes in relative resistance and relative Seebeck coefficient as evaluation parameters. The measured critical voltage values ranged from 0.02 to 0.12 V. Under constant-temperature conditions, as the amount of Cu deficiency δ increased or the ambient temperature rose, the critical voltage of the Cu2-δ(S,Se) material gradually increased, a trend that closely matched theoretical predictions, indicating that the metallic cations with "liquid-like" characteristics in the material are more difficult to precipitate. Under fixed temperature-difference conditions, the critical voltage of the Cu2-δ(S,Se) material also depended on the direction of the internal thermal flow within the material. When the direction of the thermal flow was aligned with the direction of the electric current, the material exhibited a lower critical voltage, suggesting that the metallic cations in the material were more likely to precipitate. Conversely, when the direction of the thermal flow was opposite to that of the electric current, the material showed an enhanced critical voltage, significantly improving its stability.
Based on a deep understanding of the mechanisms underlying ion migration and precipitation, the team proposed that introducing an "ion-blocking-electron-conducting" interface into quasi-liquid thermoelectric materials can effectively suppress the precipitation of metallic cations with "quasi-liquid"-like characteristics and enhance the operational stability of these materials. Since metallic cations cannot pass through the "ion-blocking-electron-conducting" interface, the external electric field will be evenly distributed among the various segments of the quasi-liquid thermoelectric material, each separated by this interface. As a result, the material as a whole can remain stable even under stronger electric fields or larger temperature differences. Meanwhile, the "ion-blocking-electron-conducting" interface does not impede the free transport of electrons and holes; thus, while achieving high operational stability, the multi-segment material still retains its intrinsic excellent thermoelectric performance. This strategy has been successfully validated in a multi-segment Cu1.97S material connected by conductive carbon layers. This work not only opens up possibilities for the practical application of quasi-liquid thermoelectric materials but also provides new insights into enhancing the operational stability of other electron-ion hybrid conductors.
The research was funded and supported by the National Key R&D Program, the National Natural Science Foundation of China, and the Youth Innovation Promotion Association of the Chinese Academy of Sciences, among others.
Link: https://www.nature.com/articles/s41467-018-05248-8

(a) The working environment of quasi-liquid thermoelectric materials; under high current conditions, (b) the precipitation of metallic Cu on the end faces of conventional quasi-liquid thermoelectric materials and (c) quasi-liquid thermoelectric materials with an "ion-blocking-electron-conducting" interface.

Physical and Chemical Processes of Ion Migration and Precipitation in Liquid-like Thermoelectric Materials

(a) Critical current and critical voltage of Cu1.97S samples with different lengths; (b) Critical voltage of Cu2-dS samples with different stoichiometric ratios; (c) Critical current of Cu1.97S samples under a given temperature difference; (d) Critical voltage of Cu1.97S under different temperature differences and heat flow directions.

The principle of enhancing operational stability by utilizing the “ion-blocking-electron-conducting” interface (a, b); (c) experimental results under constant temperature conditions and (d) under conditions with a given temperature difference.

Shanghai Institute of Ceramics Achieves Major Progress in Inorganic Plastic Thermoelectric MaterialsJanuary/08/2024

Unlock the forefront of the industry! CASBOSON invites you to the Canton Fair for in-depth exchanges.October/16/2025
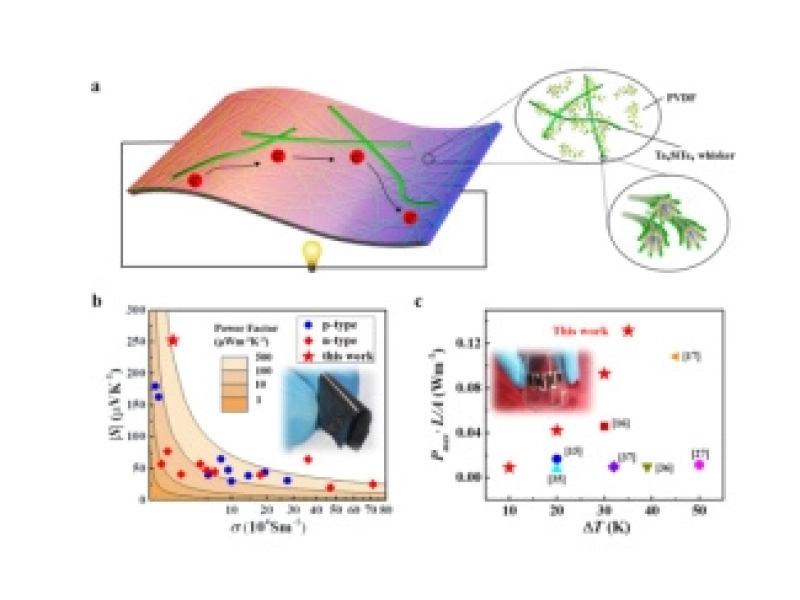
Shanghai Institute of Ceramics Achieves Major Progress in Flexible Organic/Inorganic Thermoelectric CompositesMay/26/2020
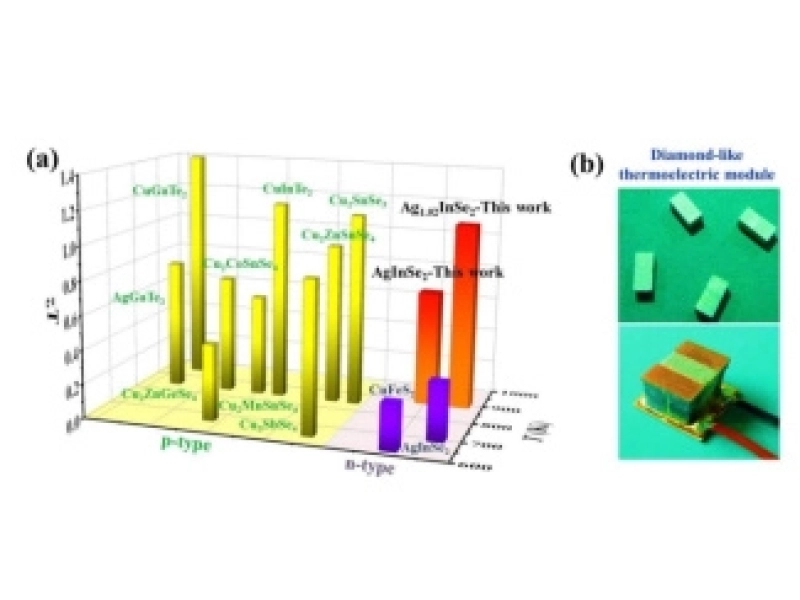
The Shanghai Institute of Ceramics has made significant progress in the research on n-type high-performance diamond-like thermoelectric materials.January/06/2018
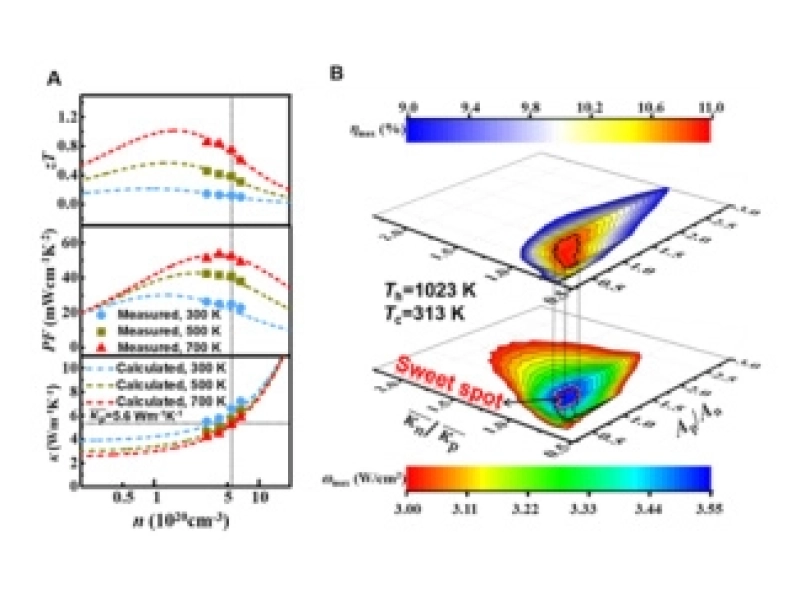
Shanghai Institute of Ceramics Achieves Progress in Thermoelectric Device ResearchSeptember/21/2020