Open Nav

Thermal conductivity is one of the fundamental physical properties of materials and plays an important, even decisive, role in many fields. Materials with high thermal conductivity are widely used in heat dissipation applications, whereas materials with low thermal conductivity are primarily employed in thermal insulation. The definition and measurement of thermal conductivity both require adiabatic conditions—no energy exchange between the material and its surroundings, meaning heat can only be conducted along the material from higher to lower temperatures. Currently, the techniques for testing thermal conductivity of materials have become quite mature. Particularly for bulk materials, international and national standards already exist for measuring thermal-conductivity-related parameters, as well as commercially available, well-established instruments. Phase transition is a characteristic exhibited by many materials. Phase-change materials find extensive applications in areas such as solid-state storage, optoelectronic switches, and energy conversion. As is well known, during a phase transition, there is significant energy exchange between the material and its environment, which becomes strongly coupled with heat transfer. Therefore, understanding and measuring the thermal conductivity of materials during phase transitions clearly differ from the situation under adiabatic conditions—a scientific issue that remains largely unknown yet is both fundamental and highly significant. Research into this issue holds promise for providing new insights and driving related applications. Especially at the current stage, numerous inconsistencies—and even completely opposing—interpretations and experimental data have emerged regarding the thermal conductivity of materials during phase transitions. For example, materials like Cu2S and Ag2S undergo first-order phase transitions; their electrical properties show no abrupt change at the phase-transition point but smoothly evolve from the low-temperature phase to the high-temperature phase. Yet, their thermal conductivities exhibit anomalous kinks, dropping below the values observed in both the low- and high-temperature phases at the transition point. Even for Cu2Se, which undergoes a second-order phase transition, the thermal conductivities calculated separately using directly measured heat-capacity values and those derived from the Dulong-Petit theoretical heat capacity display dramatically opposite trends within the phase-transition region.
Recently, Researcher Shi Xun, Researcher Chen Lidong, Researcher Zeng Huarong, and Associate Researcher Qiu Pengfei from the Shanghai Institute of Ceramics, Chinese Academy of Sciences, in collaboration with Professor Yang Ronggui from the University of Colorado/华中科技大学, clarified the impact of heat absorption and release during phase transitions on thermal transport by correcting the classical heat transport equation. They found that, in addition to affecting the material's specific heat capacity, phase transitions can also significantly give rise to an experimental artifact—namely, a reduction in the material's thermal diffusivity. The faster the phase transition occurs, the more pronounced this artifact becomes. Therefore, the true thermal conductivity of a material during phase transitions needs to account for both the additional contribution from the increased specific heat capacity and the reduced thermal diffusivity. This understanding has been experimentally validated in four phase-change materials: Cu2Se, Cu2S, Ag2S, and Ag2Se. The relevant research findings have been published in Advance Materials (DOI: adma.201806518).
At high temperatures, the thermal conductivity of a material can be calculated using the formula k = CP′d′l, where CP is the specific heat capacity, d is the density, and l is the thermal diffusivity. Typically, the laser flash analysis (LFA) method is used to measure thermal diffusivity, the Archimedes method is employed to determine density, and differential scanning calorimetry (DSC) is used to measure specific heat capacity. When a material undergoes a phase transition, it is well known that the specific heat capacity can increase significantly, while the density change is minimal and can be neglected; moreover, the thermal diffusivity is generally considered unaffected.
Material phase transitions absorb or release part of the heat, making it impossible to describe the heat flow transport during phase transitions using the classical heat conduction equation. The research team introduced a phase-transition kinetics equation for correction and successfully derived a heat conduction equation applicable to phase-transition processes. Based on this equation, they found that phase transitions can significantly affect the measurement of a material's thermal diffusivity. To characterize the relationship between phase transitions and thermal diffusivity, the team introduced the phase-transition reaction rate (B-factor). When the B-factor approaches zero, the phase transition has no impact whatsoever on the measurement of thermal diffusivity. However, when the B-factor is large, the phase transition occurs rapidly within a short time frame, exerting a substantial influence on thermal diffusivity. The research team selected four representative phase-transition materials—Cu2S, Cu2Se, Ag2S, and Ag2Se—for experimental study and characterized their phase-transition reaction rates using DSC differential scanning calorimetry. The study revealed that Cu2Se and Cu2S, with relatively large B-factors, experienced a significant reduction in thermal diffusivity during phase transitions, whereas Ag2S, with a smaller B-factor, showed only a slight decrease in thermal diffusivity during its phase transition. For Ag2Se, the B-factor was extremely low, so the phase transition had virtually no effect on thermal diffusivity.
During a phase transition, the true thermal conductivity of a material needs to account simultaneously for the removal of the extra heat capacity and the reduction in the thermal diffusivity. At high temperatures, the constant-volume heat capacity of the material is equal to the Dulong-Petit value, making it relatively easy to eliminate the added heat capacity. As for the thermal diffusivity, based on the corrected heat-transport equation, we can successfully subtract the contribution from the phase transition from the measured thermal diffusivity. Using this approach, we have successfully determined the true thermal conductivities of four materials—Cu2S, Cu2Se, Ag2S, and Ag2Se—during their respective phase transitions. In particular, the thermal conductivity of Cu2Se agrees well with values measured using both the surface heat-loss method and the 3ω method. Moreover, the corrected thermal conductivity characteristics of Cu2S and Ag2S are consistent with their electrical transport properties and the signatures of first-order phase transitions. The study also reveals that for materials with first-order phase-transition characteristics—such as Cu2S, Ag2S, and Ag2Se—the thermal conductivity shows no additional variation during the phase transition; rather, it smoothly transitions directly from the low-temperature phase value to the high-temperature phase value. However, for Cu2Se, which exhibits a second-order phase-transition characteristic, the thermal conductivity undergoes a significant decrease during the phase transition, reaching a minimum at the critical point of the phase transition. This indicates that critical fluctuations during the second-order phase transition can strongly scatter phonons, thereby reducing the thermal conductivity. Based on the calculated true thermal conductivity, the thermoelectric figure of merit of Cu2Se at the critical phase-transition point reaches 0.86—a value significantly higher than its performance before and after the phase transition. This work provides new research ideas and strategies for analyzing and controlling thermal conduction in thermoelectric materials, phase-change memory materials, solar-cell materials, and other substances undergoing phase transitions.
The research was funded and supported by the National Key R&D Program, the National Natural Science Foundation of China, the Chinese Academy of Sciences Major Scientific Equipment Development Project, and the Chinese Academy of Sciences Youth Innovation Promotion Association, among others.
Paper link: https://onlinelibrary.wiley.com/doi/10.1002/adma.201806518

(a) Specific heat and (b) thermal diffusivity of representative phase-change materials Cu2Se, Cu2S, Ag2S, and Ag2Se; (c) thermal conductivity of Cu2Se calculated based on different specific heats; (d) schematic diagram illustrating the impact of heat absorption and release during the phase-change process on heat transfer.

(a) The relationship among the thermal diffusivity (lm), phase transition reaction rate, and specific heat during the phase transition process; (b) The variation of phase transition reaction rates of Cu2Se, Cu2S, Ag2S, and Ag2Se with temperature.

(a) The actual thermal conductivities of Cu2Se, Cu2S, Ag2S, and Ag2Se; (b) The thermoelectric figure of merit zT calculated based on the actual thermal conductivities during the phase transition of Cu-2Se.
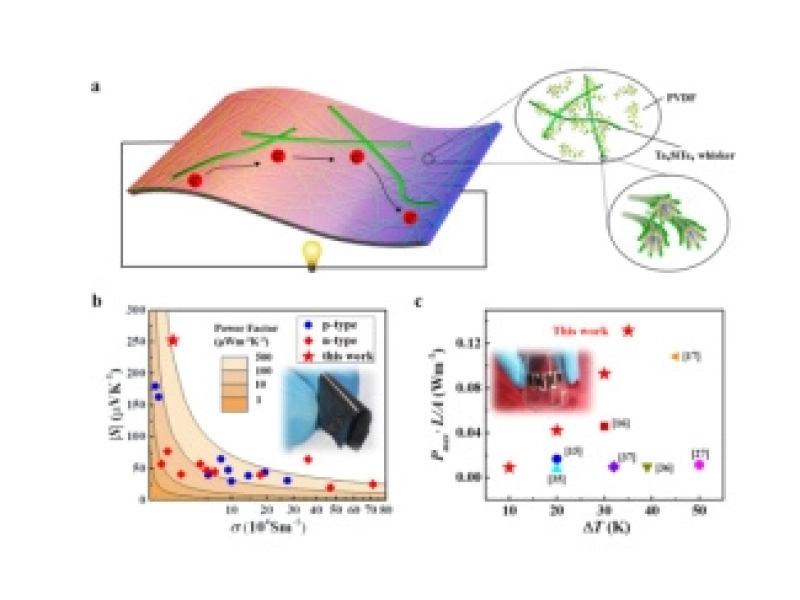
Shanghai Institute of Ceramics Achieves Major Progress in Flexible Organic/Inorganic Thermoelectric CompositesMay/26/2020
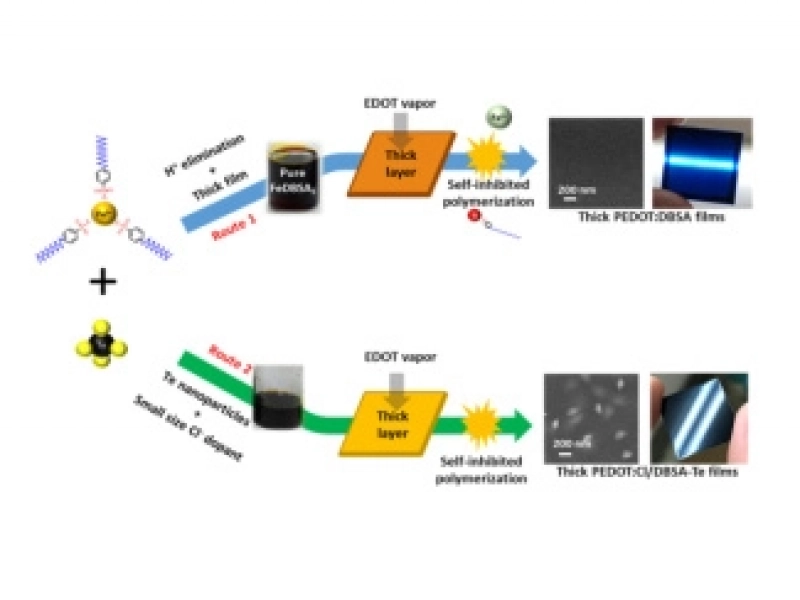
The Shanghai Institute of Ceramics has made significant progress in the field of organic-inorganic composite thermoelectric materialsJuly/17/2018

Shanghai Institute of Ceramics Hosts 2022 Annual Exchange and Sharing Meeting for SICCAS Holdings Management Company Portfolio EnterprisesJanuary/25/2023

Unlock the forefront of the industry! CASBOSON invites you to the Canton Fair for in-depth exchanges.October/16/2025
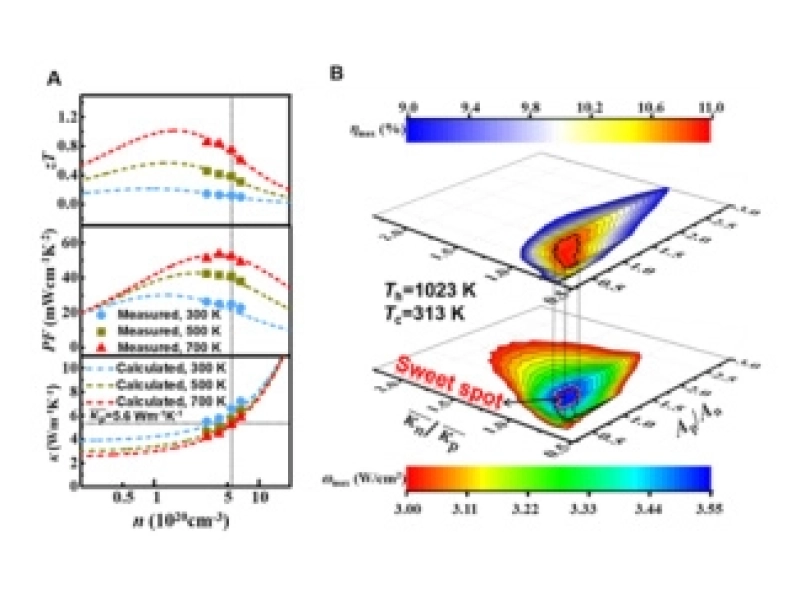
Shanghai Institute of Ceramics Achieves Progress in Thermoelectric Device ResearchSeptember/21/2020